 In our patient, the development of hematuria may result from the anti-VEGF effect of bevacizumab. Inhibition of VEGF, which has anti-apoptotic, anti-inflammatory, and pro-survival influences on endothelial cells, weakens their regenerative capacity and increases expression of proinflammatory genes leading to weakened supporting layers of blood vessels and, hence, to damaged vascular integrity. The patient was a cervical cancer with bladder metastasis that increases the risk of development of hematuria. After stopping chemotherapy, cefoxitin, tranexamic acid and hemocoagulase atrox therapy was administered resulting in rapid clinical improvement. Manifested as massive gross hematuria with blood clots. After three cycles every three weeks of consolidation chemotherapy (carboplatin, paclitaxel, bevacizumab) and following three cycles consolidation chemotherapy (carboplatin, paclitaxel, bevacizumab, pembrolizumab), the patient presented a worsening state. We report a severe hematuria of bevacizumab plus pembrolizumab, in a 58-year-old woman with metastatic cervical cancer. By blocking the activity of PD-1, the purpose of inhibiting tumor growth is achieved. 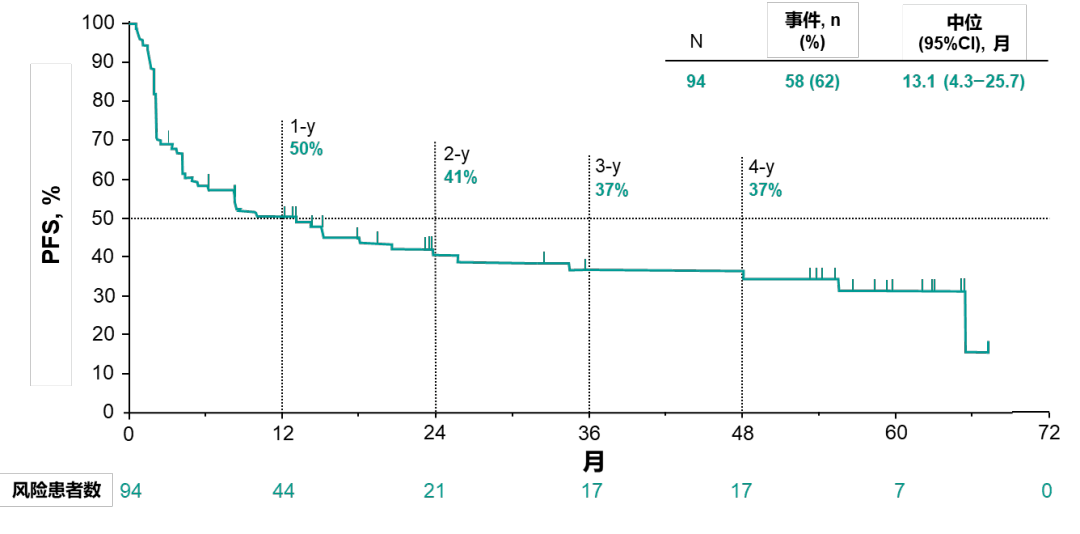
Pembrolizumab is a monoclonal antibody that can bind to the programmed death-1 (PD-1) receptor, which can block the binding of the PD-1 receptor to its ligands PD-L1 and PD-L2, and release PD-1 pathway-mediated suppression of immune responses. SGO 2021 Abstract 10417.Bevacizumab is a monoclonal antibody drug targeting Vascular Endothelial Growth Factor (VEGF), which binds to VEGF receptors to inhibit vascular endothelial cell proliferation and angiogenesis, thus inhibiting tumorigenesis. Interim analysis of the immune-related endpoints of the mismatch repair deficient (dMMR) and proficient (MMRp) endometrial cancer cohorts from the GARNET study. Pembrolizumab in patients with MSI-H advanced endometrial cancer from the KEYNOTE-158 study. Clinical activity and safety of the anti-programmed death 1 monoclonal antibody dostarlimab for patients with recurrent or advanced mismatch repair-deficient endometrial cancer: A nonrandomized phase 1 clinical trial. ENGOT-EN6/NSGO-RUBY: A phase III, randomized, double-blind, multicenter study of dostarlimab + carboplatin-paclitaxel versus placebo + carboplatin-paclitaxel in recurrent or primary advanced endometrial cancer (EC). Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer. A multicenter, open-label, randomized, phase III study to compare the efficacy and safety of lenvatinib in combination with pembrolizumab versus treatment of physician’s choice in patients with advanced endometrial cancer: Study 309/KEYNOTE-775. 
Manage wisely: poly (ADP-ribose) polymerase inhibitor (PARPi) treatment and adverse events. Talazoparib in patients with advanced breast cancer and a germline BRCA mutation. The use of pembrolizumab and lenvatinib combination therapy in endometrial cancer: An examination of toxicity and treatment efficacy in clinical practice. Tisotumab vedotin in previously treated recurrent or metastatic cervical cancer. Tisotumab vedotin in previously treated recurrent or metastatic cervical cancer: Results from the phase II innovaTV 204/GOG-3023/ENGOT-c圆 study. SGO 2021 Abstract 10440.Ĭoleman RL et al. Pembrolizumab treatment of advanced cervical cancer: Updated results from the phase II KEYNOTE-158 study. 
Maintenance olaparib for patients (pts) with newly diagnosed, advanced ovarian cancer (OC) and a BRCA mutation (BRCAm): 5-year (y) follow-up (f/u) from SOLO1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
